Despite over $60 billion in venture capital pouring into AI-driven drug discovery since 2015 and myriad computational breakthroughs, as of 2025 not a single AI-designed drug has achieved FDA approval. Nor has any received a positive reimbursement recommendation from a health technology assessment (HTA) body. These stark outcomes highlight a widening disconnect between Silicon Valley’s “move fast and break things” ethos and the sobering realities of bringing safe, effective, and cost-effective medicines to patients.
The industry now faces a crucial inflection point: early promises of dramatic efficiency gains have not translated into clinical success, and a critical bottleneck in market access and reimbursement remains largely ignored. This deep-dive analysis examines how lofty expectations have collided with biological complexity and commercial realities – and what it will take to bridge the gap between AI’s potential and tangible patient impact.
The Promise vs. Performance Paradox
The AI drug discovery sector has attracted extraordinary investment based on compelling narratives of transformation. Since 2015, over 800 AI-driven pharmaceutical companies have secured funding, with individual raises reaching unprecedented levels (e.g. Xaira Therapeutics secured $1 billion without any drug in trials). In 2024 alone, the sector attracted $5.6 billion in new VC funding. The narrative was seductive: AI algorithms would revolutionize drug discovery, reduce drug development timelines from 12-15 years to under 5 years, slash costs from $2.5 billion to under $500 million per approved drug, and improve clinical success rates from 10% to potentially 90%.
The actual performance tells a different story. Of approximately 75 AI-discovered molecules that have entered human trials since 2015, none have progressed beyond Phase II clinical trials. While AI-designed drugs show promising Phase I success rates of 80-90% compared to 40-65% for traditional approaches, this advantage disappears in Phase II, and so far, only a handful of AI-designed drugs have managed to enter Phase III:
RLY-2608 stands alone as the first AI-designed drug to enter Phase III trials. The ReDiscover-2 trial, initiated in Q2 2025, compares RLY-2608 plus fulvestrant against capivasertib plus fulvestrant in HR+/HER2- advanced breast cancer patients with PIK3CA mutations. This represents a major milestone for the field.
Rentosertib appears poised to be the second, with Insilico actively engaging regulatory authorities for a potentially pivotal trial in H2 2025. The company's CEO stated they will be "pursuing a potentially pivotal trial of ISM001-055 in IPF patients," with a planned global Phase IIb study enrolling approximately 270 patients.
TAK-279 (originally Nimbus Therapeutics' NDI-034858) represents a special case - while AI-assisted in development, it was acquired by Takeda and is now in multiple Phase 2/3 studies for psoriasis and psoriatic arthritis, though it's not purely AI-designed in the same sense as Insilico's or Relay's drugs.
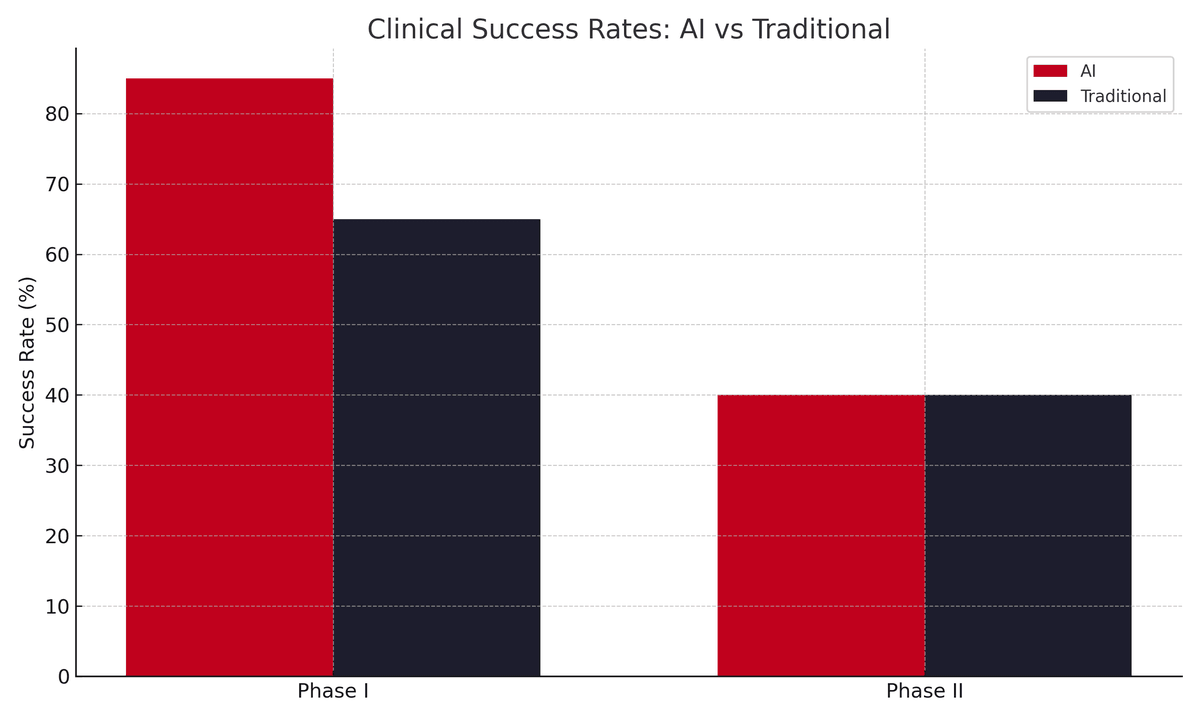 Clinical trial success rates for AI-designed vs. traditionally discovered drug candidates. AI approaches show markedly higher Phase I success (reflecting strong early safety/PK profiles), but by Phase II their success rate falls in line with industry averages.
Clinical trial success rates for AI-designed vs. traditionally discovered drug candidates. AI approaches show markedly higher Phase I success (reflecting strong early safety/PK profiles), but by Phase II their success rate falls in line with industry averages.
The financial mathematics are sobering. With $60 billion invested across approximately 150 drug candidates in development, the current investment per clinical candidate exceeds $400 million - before any have reached market. For comparison, the traditional pharmaceutical industry's often-criticized $2.5 billion per approved drug includes the full cost of failures, while AI's current track record shows only costs without approvals. As Patrick Malone of KdT Ventures noted, “If you took the hype at face value these past 10 years, you’d think success rates went from 5% to 90%. But if you know how these models work, it’s more like 5% to maybe 6–7%.” In other words, the actual boost from AI might be incremental – not revolutionary – in the near term.
This can be visualized by plotting the cumulative number of AI-designed drug candidates entering trials versus those reaching approval. The pipeline has expanded rapidly in recent years as dozens of AI-derived compounds enter Phase I/II, but approvals remain a flat zero:
High-profile failures expose fundamental challenges
A series of high-profile clinical setbacks have tempered early optimism and provided case studies in AI’s current limitations in drug discovery. Exscientia, despite being first to bring an AI-designed drug to human trials in 2020, has seen multiple programs fail or be discontinued. Their DSP-1181 for obsessive-compulsive disorder, developed with Sumitomo Pharma, was abandoned in Phase I after failing to meet study criteria, Its cancer immunotherapy EXS-21546 was winded down in 2023 when models suggested the required therapeutic index would be “challenging” – an admission that computational predictions failed to anticipate biological reality. The company’s once high-flying stock collapsed ~79% from its peak, and in August 2024 Exscientia agreed to a fire-sale acquisition by Recursion for $688 million in stock (a fraction of its earlier >$2 billion valuation).
BenevolentAI's experience proves even more sobering. Their topical pan-Trk inhibitor BEN-2293 for atopic dermatitis met safety requirements but showed no statistically significant efficacy versus placebo in Phase IIa trials. This failure triggered massive layoffs, with the company cutting 180 staff in May 2023 and an additional 30% and closed its US office in 2024, while its stock price plummeted over 75% from peak valuation.
Perhaps most dramatically, IBM Watson for Drug Discovery - launched with fanfare and partnerships with Pfizer, Johnson & Johnson, and Sanofi - was shut down entirely in April 2019 after "lackluster financial performance." IBM had invested an estimated $5 billion in Watson Health acquisitions only to sell the assets for approximately $1 billion, a massive destruction of value that Derek Lowe, a pharmaceutical researcher, attributed to the system simply not being "ready for those challenges."
Recursion Pharmaceuticals, despite maintaining the highest valuation among first-generation AI biotechs at over $1 billion, reduced its pipeline from 11 to 6 active programs in December 2024. Their discontinued programs include treatments for rare brain diseases, neurofibromatosis, and C. difficile infection. Recursion’s Phase II program REC-994 (for cerebral cavernous malformation) initially showed encouraging MRI biomarker changes, but longer-term data found it yielding no distinguishable patient benefits. The program was discontinued, contributing to Recursion’s hefty $463.66 million net loss in 2024 (41% deeper than 2023) despite $58.8 million in sales revenue.
Perhaps most sobering is the perspective of insiders who helped invent the field. Brendan Frey, founder of Deep Genomics (and co-founder of the Vector Institute), admitted in 2024 that “AI has really let us all down in drug discovery… we’ve seen failure after failure.” His company, despite ~$250 million raised, was reported to have a “flailing” pipeline and was exploring a sale. Such frank admissions from AI drug discovery’s earliest champions signal a broader reckoning: the field’s short-term promises have been severely overestimated.
Collectively, these setbacks illustrate that AI, in its current form, cannot eliminate the fundamental risks of drug R&D. Even when AI churns out a plausible molecule that performs in silico and in animal models, human biology often has the last word – and it’s often “no.” The failures also highlight the human cost of hype cycles: hundreds of talented scientists laid off, years of work and capital evaporated, and patients still waiting for new treatments that didn’t materialize.
The Computational–Biological Translation Gap
The core challenge facing AI drug development lies not in computational capability but in biological complexity. As DeepMind’s Demis Hassabis noted, “Biology is likely far too complex and messy to ever be encapsulated as a simple set of neat mathematical equations.” This fundamental reality manifests in multiple ways that current AI systems struggle to address.
First, there's the "black box" problem - while AI can identify patterns in data, it often cannot fully explain why certain molecules might work, making it difficult to optimize candidates when they fail. Harvard's Wyss Institute research highlights that generative AI "often suggests compounds that are challenging or impossible to synthesize or lack drug-like properties"- a modern version of what Nature calls the "ChatGPT problem" in drug discovery.
Second, data quality issues plague the field. AI models are only as good as their training data, and pharmaceutical datasets suffer from publication bias (negative results rarely published), incomplete safety information, and overrepresentation of certain populations and experimental conditions. As Alex Zhavoronkov, founder of Insilico Medicine, warns, the industry has created "dangerous financial hype" with companies receiving funding companies based on promises and big names rather than validated milestones.
The validation gap proves particularly troubling. Research by Scannell & Bosley demonstrates that a 0.1 absolute change in correlation coefficient between model output and clinical outcomes can offset 10-100 fold changes in screening efficiency. In other words, predictive validity matters far more than computational speed - yet the industry has progressively abandoned high-validity models for low-validity, high-throughput approaches.
Market Access: the overlooked Valley of Death
Even if AI-designed drugs overcome clinical hurdles, they face a second, often-overlooked challenge: market access and reimbursement. Research reveals that 40% of drugs approved between 2004-2016 underperformed Wall Street forecasts, with the average number of patients per launch brand in the first year dropping from 180,000 in 2007 to just 42,000 in 2016.
The reimbursement infrastructure presents formidable barriers. In the United States, drugs face an average 9.2 months between FDA approval and reimbursement decisions, while in England this extends to 17.7 months. Health Technology Assessment (HTA) bodies like NICE in the UK and ICER in the US demand extensive evidence of cost-effectiveness, with thresholds of $50,000-100,000 per quality-adjusted life year (QALY) that many innovative drugs struggle to meet.
Current HTA frameworks lack specific guidance for assessing AI-designed drugs, with research showing that AI studies achieve only a 52% average HTA score across evaluation domains. Only 9% of AI-based medical device studies evaluate safety aspects adequately, while just 20% address economic considerations. This creates a paradox: AI drug developers focus on computational innovation and regulatory approval while neglecting the market access considerations that determine commercial viability.
The disconnect is striking. A Value in Health paper shows that 79% of US payers are influenced by ICER recommendations, yet most AI drug companies keep reimbursement teams "on the sidelines until Phase III" or later. Companies typically begin market access planning only 6-18 months before launch instead of the recommended 18-24 months, missing critical opportunities to generate payer-relevant evidence during development.
Regulatory frameworks need to keep pace
The FDA has been proactive in addressing AI in drug development, releasing its first-ever draft guidance in January 2025 titled "Considerations for the Use of Artificial Intelligence to Support Regulatory Decision-Making for Drug and Biological Products." Since 2016, the agency has reviewed over 500 drug submissions with AI components, with more than 100 in 2021 alone.
However, significant challenges remain. The FDA requires comprehensive validation demonstrating model performance for specific contexts of use, yet the "black box" nature of many AI systems makes this transparency difficult to achieve. Data quality and bias concerns persist, particularly around ensuring training data representativeness and managing historical biases in clinical trial data.
While companies like Insilico Medicine have achieved 10 IND (Investigational New Drug) approvals for AI-designed compounds, with their INS018_055 for idiopathic pulmonary fibrosis receiving Orphan Drug Designation and entering Phase 2 trials, the earliest potential FDA approvals for AI-designed drugs remain 2-5 years away. The regulatory timeline from IND to approval typically spans 7-9 years, meaning even the most advanced AI candidates won't reach market before 2027-2028 at the earliest.
The implication is multifaceted: AI-driven biotechs must integrate market access early or risk a “valley of death” after approval. This means conducting payer-relevant studies (e.g. comparing to standard of care, collecting patient-reported outcomes), seeking dialogues with HTA bodies, and devising pricing models (outcomes-based, staggered payments, etc.) if pursuing ultra-expensive cures. Notably, startups are tackling this infrastructure gap, with Loon developing AI-powered platforms to streamline clinical research, optimize drug reimbursement submissions, and model various reimbursement scenarios at scale. Such tools help bridge the knowledge gap for biotechs not yet versed in health economics. Ultimately, success in the market will demand the same data-driven rigour as R&D: evidence of comparative effectiveness and value for money.
The venture capital hype machine
The investment patterns in AI drug discovery reveal a classic venture capital hype cycle. The VC formula has become predictable: identify a trend, craft a compelling narrative, assemble prestigious names, and syndicate substantial raises. This "faith-based" approach, as industry observers describe it, has led to massive valuations disconnected from clinical reality. All major first-generation AI biotechs have seen their stocks decline 75% or more from peak valuations, with BenevolentAI falling from over $1 billion to approximately $117 million.
Jessica Owens, co-founder of Initiate Ventures, identifies a critical misalignment: VCs historically avoid service companies due to margin and scalability concerns, preferring "hockey stick" growth narratives over steady, proven business models. This bias toward "glamorous" drug discovery — over necessary but the less glamorous infrastructure work like evidence synthesis and market access planning — has contributed to the sector's struggles.
The concentration of investment proves telling. The top 20 AI-first biotechs have captured 60% of total investment, with 76% of funding concentrated in the United States. Yet this concentration hasn't translated into success - these companies collectively show accumulated deficits exceeding $1.5 billion with zero approved drugs to show for it.
The methodical alternative: what actually works
Against this backdrop of hype and failure, research consistently points to methodical, evidence-based approaches as the key to successful drug development. AstraZeneca's analysis of 150+ drug projects identified five critical success factors that have nothing to do with computational speed:
right target (clinically validated),
right tissue (demonstrated engagement),
right safety (adequate margins),
right patients (biomarker-guided),
right commercial potential (clear value proposition).
The FDA's Process Validation framework, refined over decades, emphasizes three stages that cannot be accelerated by drug discovery computation alone: Process Design (defining the commercial process), Process Qualification (evaluating performance), and Continued Process Verification (ongoing monitoring). These requirements, spanning 12-15 years and costing $1-2.6 billion per approved drug, reflect biological and regulatory realities that persist regardless of how drugs are discovered.
Bridging the Gap from Hype to Impact
The AI drug discovery sector stands at a critical juncture. After a decade of promises and $60 billion in investment, the industry must confront an uncomfortable truth: computational capability alone cannot overcome biological complexity. The persistent 90% failure rate in clinical development, despite technological advances, underscores that predictive validity - not computational speed - remains the primary constraint on pharmaceutical R&D productivity.
The path forward requires abandoning Silicon Valley's "move fast and break things" mentality in favour of the methodical, evidence-based approaches that have historically delivered safe and effective medicines. According to McKinsey & Company, this means:
accelerating clinical and evidence synthesis research
accelerating AI clinical (not drug) development
market access operations
commercialization
medical affairs
For investors, the lesson is clear: sustainable returns in drug development come not from betting on drug development computational promises but from backing companies that combine AI capabilities with deep clinical expertise, rigorous validation processes, and comprehensive market access strategies. The companies that succeed will be those that use AI to enhance, not replace, the fundamental disciplines of pharmaceutical development.
The ultimate measure of success in drug development remains unchanged: delivering safe, effective, and accessible treatments to patients. Until AI-designed drugs achieve this goal, the $60 billion invested represents not a revolution but an expensive education in the enduring challenges of pharmaceutical innovation.
Navigate the Complexities of Market Access with Expert Insights
Learn how Loon's evidence-based solutions can help accelerate your HTA submissions and market access strategies.
Schedule a ConsultationFrequently Asked Questions
Frequently Asked Questions
Start Transforming Your HTA and Market Access Strategy Today
Join pharmaceutical companies that are accelerating their market access with evidence-based AI solutions.
Schedule Your Consultation